
After obtaining the density function, I normalized it across an arbitrary distance between two parallel planes equidistant from the nucleus so that the planes enclosed 5 of the 6 electrons. 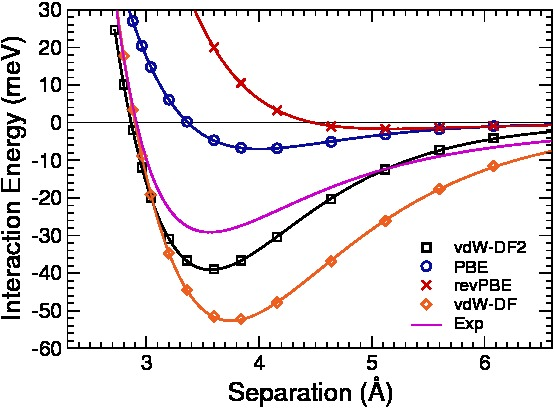
I then determined wave functions for each orbital in the structure, and wrote the electron density as an appropriate linear combination of their magnitudes. In my model, I assumed that each carbon atom had the electron configuration: 1s^22s2p^3 (4 valence electrons). I have implored a specific model in density functional theory called the Thomas-Fermi-Dirac Method, along with a correction for the inhomogeneity of kinetic energy that was calculated by Kirzhnits, to predict the Van der Waals forces between layers in a graphite crystal. There are many variations of density functional theory, along with many corrections made to each. 
AbstractDensity functional theory is an approach to mathematically model and interpret complex many-atom systems by expressing the energy of the system as a functional of electron density.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
